Faith Nyasuguta
In Kenya, over 3.5 million people get malaria every year, according to CDC estimates. Out of the numbers, over 10,000 Kenyans die from it every year.
According to Francis Ndungu, a malaria immunologist at KEMRI-Wellcome Trust: “In the case of malaria, a mosquito bites you and injects a parasite form called sporozoites into your skin and those sporozoites through the lymph and the blood circulatory system and eventually ends up in the liver. So the vaccine that we are talking about, R21/Matrix-M vaccine, interferes with the sporozoites, stopping them from invading the liver cells.”
Hope is on the horizon with a new vaccine, R21, whose preliminary results from early testing have suggested that it is far more effective than the RTS,S malaria vaccine, the only one currently authorised for use by the WHO.
The final results from the late-stage trials have not yet been published, and the R21 vaccine is under review at the World Health Organization.
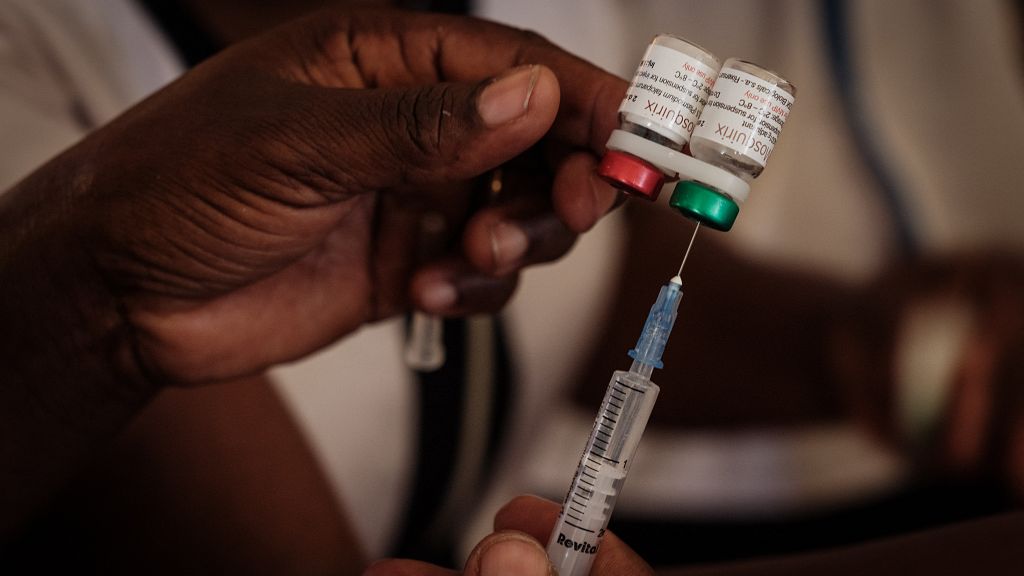
On April 13, Ghana became the first country to approve its rollout. Nigeria has also given a nod to its rollout.
The WHO has already rolled out a pilot program of the world’s first authorised malaria vaccine known as RTS,S, piloted in three African countries, including Kenya. But it’s not widely available yet.
The efficacy of the R21 vaccine is 75% compared with the RTS,S vaccine, sold by GlaxoSmithKline as Mosquirix, which is about 30% effective.
The RTS,S malaria vaccine came as a result of three decades of research and development by GSK. It was approved by the WHO in 2021.
“This vaccine is not perfect so in fact RTS,S offers limited protection that is also short lived,” says Francis Ndungu, a malaria immunologist at KEMRI-Wellcome Trust.
The scientists at the KEMRI-Wellcome Trust in Kenya are involved in the R21 trial and Ndungu is the lab’s principal investigator for the project.
He says: “the WHO benchmark for a good vaccine to aim for is 75 percent and above protection from clinical malaria and so we are very excited about the R21/Matrix-M vaccine because it is actually the first vaccine that has exceeded the WHO benchmark for a highly effective malaria vaccine.”

Ndungu says the ongoing phase three trials involving 4,800 children will provide crucial data to WHO:
” With the data from the ongoing phase 3 trial in Kenya, Tanzania, Mali and Burkina Faso, once that data is provided to WHO. WHO will also evaluate that data and if they are convinced that the vaccine is safe and highly protective, then they will give provisional approval for the use of that vaccine for wider use in more territories,”
Ndungu says malaria not only takes lives it destroys communities.
“Apart from death, disease also interferes with people’s lives, education, economic activities and also the societies economy and so it is very important to a highly effective anti-malarial vaccine that will save children from death and also give them the opportunity to develop, grow and develop in the absence of disease so that they do better in school and also in life,” he says.
According to the World Health Organization, 247 million people had malaria in 2021 and that year it killed 619,000 people.
The vast majority of malaria cases are on the African continent, 96 percent of all the deaths in this region are among children under the age of five.
Malaria is mostly found in tropical countries and is spread to humans by the female Anopheles mosquito.
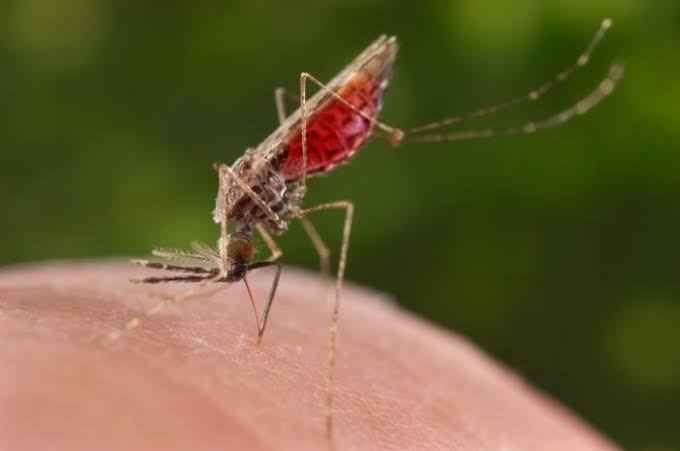
The life threatening disease has symptoms such as fever, chills, headaches, fatigue, confusion, seizures and difficulty breathing.
Pregnant women, children under 5 years, people with HIV/AIDS are at high risk of infection.
The disease can be prevented with medicine and by avoiding mosquito bites through measures such as sleeping under a treated mosquito net.




