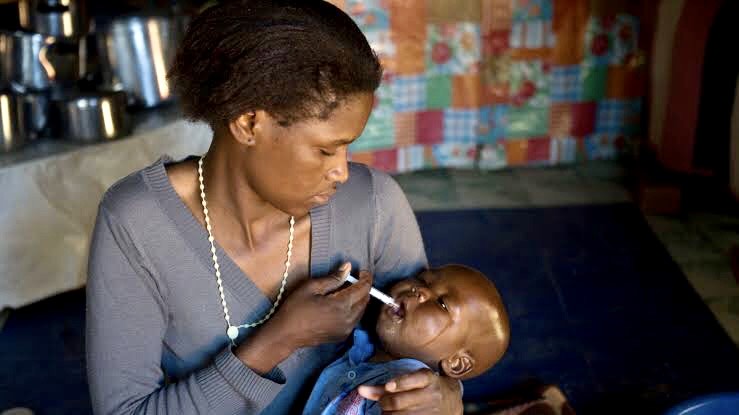
By Faith Nyasuguta
After years of waiting, Kenya and Uganda have finally introduced a strawberry-flavored tablet for children living with the human immunodeficiency virus (HIV).
The pediatric formulation of dolutegravir, a drug recommended as first-line anti-retroviral for HIV treatment by the World Health Organisation (WHO), has been lauded as a game-changer in managing the virus in children as it eases treatment, making it cheaper.
The new drug is in the form of a tablet that will be dissolved in water or juice, making it easier to give to children too young to swallow tablets.
Kenya and Uganda are among the six African countries picked to receive the dolutegravir (pDTG), which comes in formulations of 10 milligrams.
The other nations set to receive donations of up to 100,000 packs of the new drug by the Unitaids, a global health agency, and Clinton Health Access Initiative (CHAI) are Benin, Malawi, Nigeria, and Zimbabwe.

DEATH RATES
The first-line HIV treatment is endorsed by the World Health Organisation from the age of a month and from the weight of three kilograms.
However, the drug has been out of reach of children following the lack of required formulations.
Pediatric HIV experts attest that the added flavor now renders the drug more palatable to children when compared to the other available options that are bitter to the taste or even hard for babies to ingest.
Globally, some 1.8 million live with HIV but only 50 per cent receive any treatment. This is because the treatment is often hard to administer over the bitter taste and incorrect dose that arises from crushing adult pills.
Annually, approximately 100,000 children die from Aids-related causes.

Unaids says that treatment coverage among children living with HIV lags behind adult treatment coverage in the majority of the sub-Saharan African countries with large HIV epidemics.
Additionally, studies show that a huge chunk of people enrolled in treatment have family members, among them children, whose HIV status is unknown.
“Treatment coverage among children living with HIV in 2019 was just 53 percent, representing a global failure to provide life-sustaining care to 840,000 of the 1.8 million children living with HIV,” the UN agency notes.
Currently, Kenya, which received 47,000 packs of Dolutegravir (DTG) 50mg donated to the Ministry of Health by the United Nations and 28,000 packs of Dolutegravir 10mg from Unitaid through the CHAI, is implementing a seasoned transition of children and adolescents living with HIV to optimal regimens.
The phased transition from the use of Nevirapine to Efavirenz then to (DTG) 50 mg and now to DTG 10mg was launched in October 2019.
The health ministry says that the program has transitioned over 65 per cent of children on the first-line Antiretroviral Therapy (ART) to DTG-based regimens.
“Data shows that the lowest suppression is amongst the younger children. This can be attributed to several factors but key is that some of the ARVS for children, such as Lopinavir solution, have challenges of storage, palatability as well as dosing and administration,” Dr. Catherine Ngugi, who heads the National Aids and STIs Control Programme (Nascop) said.
“The availability of DTG 50mg film-coated tablets will foster improved adherence among the Children and Adolescents Living with HIV thereby improving treatment outcomes in children and adolescents,” Dr. Mercy Mwangangi, the chief administrative secretary at the ministry said.
In both the low- and middle-income nations, the pediatric formulation of dolutegravir will be priced at $36 per child per year, in line with the pricing agreement brokered by Unitaid and Clinton Health Access Initiative with generic manufacturers Viatris and Macleods.





1 Comment
I commend the thought of making medicine palatable to children. Taking a drug as a child wasn’t easy. May more of these come by to ensure good health of more and more children